Abstract
Background: Prior work has shown that clonal hematopoeisis of indeterminate potential (CHIP) is present in 12% of 285 patients with chronic lymphocytic leukemia (CLL) (median age 71 years) (Br J Haem. 2022;198:103). Additionally, CHIP was common (19%) among 89 patients with relapsed/refractory CLL exposed to venetoclax (Blood 2022;139:1198). Beyond these data, CHIP in patients with CLL has not been extensively characterized. The objective of this study is to describe the prevalence of myeloid CHIP (M-CHIP) in patients with CLL, and to determine the association of M-CHIP with overall survival (OS), time to first treatment (TTFT), and incidence of Richter's syndrome (RS).
Methods: We retrospectively analysed data from 975 patients from a prospective CLL tissue bank and database at Dana Farber Cancer Institute and who had a targeted 95-gene next-generation sequencing (NGS) panel performed as part of standard of care (J Mol Diagn. 2016;18:507). We utilized published classification criteria for M-CHIP to create a list of 56 candidate genes (Nat Med. 2021;27:1921). Given the overlap between M-CHIP and CLL driver genes, we excluded overlap genes that were more common in CLL than M-CHIP (SF3B1, TP53, BRAF, KRAS, BCOR, NRAS, CREBBP, SETD2; Knisbacher. Nat Genetics. In press). We analysed CHIP as a binary (present/absent) and categorical (>2 vs. 1 vs. 0 mutations) predictor. We controlled for the covariates of age at time of NGS panel and prior treatment; other covariates considered included IGHV and 17pdel/TP53 mutation status, but these did not differ between groups. We performed landmark analyses from time of NGS panel to assess the association of M-CHIP with OS, TTFT, and RS. We computed unadjusted and adjusted hazard ratios with 95% confidence intervals for the outcomes of interest.
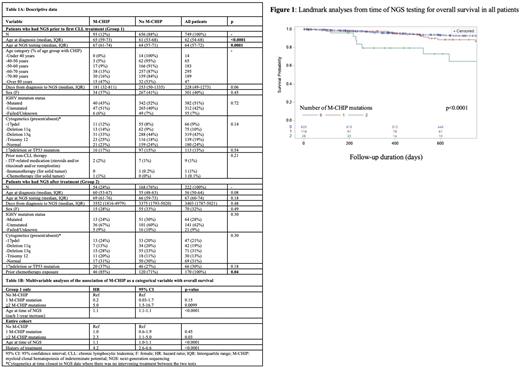
Results: Of the 975 patients assessed, 749 (77%) had NGS performed prior to CLL treatment (Group 1), while 226 (23%) had it performed after receiving treatment (Group 2). NGS was primarily performed on peripheral blood (96%; 4% on bone marrow). Median follow-up time was 700 days (interquartile range (IQR) 200-1246). Median age at time of NGS was 65 years (IQR 58-72) and 40% of patients were female (Table 1A for patient characteristics). The prevalence of M-CHIP was 12% (N=93) in Group 1, and 24% (N=54) in Group 2. The prevalence of M-CHIP appeared to increase by age in Group 1, but appeared consistent across age groups in Group 2 (by decile of age from <40 to >80; Group 1: 0%, 5%, 9%, 13%, 16%, 47%, p<0.0001; Group 2: 33%, 29%, 17%, 22%, 32%, 26%, p=0.6). Patients with M-CHIP in Group 1 were older than patients without M-CHIP in Group 1, while patients with M-CHIP in Group 2 were more likely to have previous chemotherapy/chemo-immunotherapy exposure (p=0.04), but there were no other significant differences between groups (Table 1A).
In Group 1, CHIP as a categorical predictor (>2 mutations), but not a binary predictor, was associated with OS in both univariable and multivariable analyses (Table 1B, p=0.0099) but not associated with TTFT as either a binary or categorical predictor. In the entire cohort, CHIP as a categorical predictor (>2 mutations) was associated with OS in univariable and multivariable analyses (Table 1B, Figure 1, p=0.03). Among both Group 1 and the entire cohort, CHIP as a binary predictor was not associated with Richter's syndrome (N=28 events). In the entire cohort, CHIP as a categorical predictor (>2 mutations) was associated with Richter's syndrome in univariable analysis, but this association did not hold in multivariable cause-specific hazard analysis considering death as a competing event.
Conclusions: In our cohort, the prevalence of M-CHIP in untreated (12%) and treated (24%) patients with CLL was similar to previous literature. M-CHIP prevalence appeared to increase with age in untreated patients, but appeared consistent across age groups in treated patients, suggesting that treatment (85% had prior chemotherapy) may have impact on M-CHIP emergence even in younger patients. The presence of two or more M-CHIP mutations in all patients was associated with OS, even accounting for prior treatment and age, but was driven by a small subset of patients (N=28). M-CHIP did not appear to be associated with TTFT or RS, but the latter analysis is limited in power due to few RS events. Further analyses are ongoing to assess whether increased deaths in patients with multiple M-CHIP mutations were related to non-CLL causes.
Disclosures
Getz:MSIDetect: Patents & Royalties; IBM: Research Funding; MSMuTect: Patents & Royalties; Scorpion Therapeutics: Consultancy, Current equity holder in publicly-traded company, Other: Founder; POLYSOLVER: Patents & Royalties; Pharmacyclics: Research Funding; SignatureAnalyzer-GPU: Patents & Royalties; MSMutSig: Patents & Royalties. Wu:BioNTech: Current equity holder in publicly-traded company; Pharmacyclics: Research Funding. Davids:Ono Pharmaceuticals: Consultancy; BeiGene: Consultancy, Membership on an entity's Board of Directors or advisory committees; Bristol-Myers Squibb: Consultancy, Membership on an entity's Board of Directors or advisory committees; Takeda: Consultancy; AstraZeneca: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Ascentage Pharma: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; AbbVie: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Travel expenses, Research Funding; Adaptive Biotechnologies: Consultancy, Membership on an entity's Board of Directors or advisory committees; Merck: Consultancy; Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees; Research to Practice: Honoraria; TG Therapeutics: Consultancy, Research Funding; Novartis: Research Funding; Eli Lilly and Company: Consultancy, Membership on an entity's Board of Directors or advisory committees; Genentech: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Verastem: Consultancy, Research Funding. Brown:Abbvie: Consultancy; Gilead: Research Funding; SecuraBio: Research Funding; Acerta/ Astra-Zeneca: Consultancy; MEI Pharma: Consultancy, Research Funding; TG Therapeutics: Research Funding; Genentech/Roche: Consultancy; Beigene: Consultancy, Research Funding; Bristol-Myers-Squibb/Juno/Celgene: Consultancy; Catapult: Consultancy; Eli Lilly: Consultancy, Research Funding; Sun: Research Funding; Pharmacyclics: Consultancy; Janssen: Consultancy; iOnctura: Consultancy; Hutchmed: Consultancy; Grifols: Consultancy; Pfizer: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal